MoCRA compliance does not have to delay your launch. BeyondBound provides clear, guided support for FDA facility registration and cosmetic product listings. Designed for emerging and established cosmetic brands.
MoCRA filings can feel confusing and time-consuming, especially if you’re working through them for the first time.
Included with every filing are confirmation evidence, a share-ready Proof Bundle, a filing tracker, and an update calendar that supports biennial facility renewals and annual product listing updates.
We Support:
Reduce avoidable back and forth and move forward with confidence.

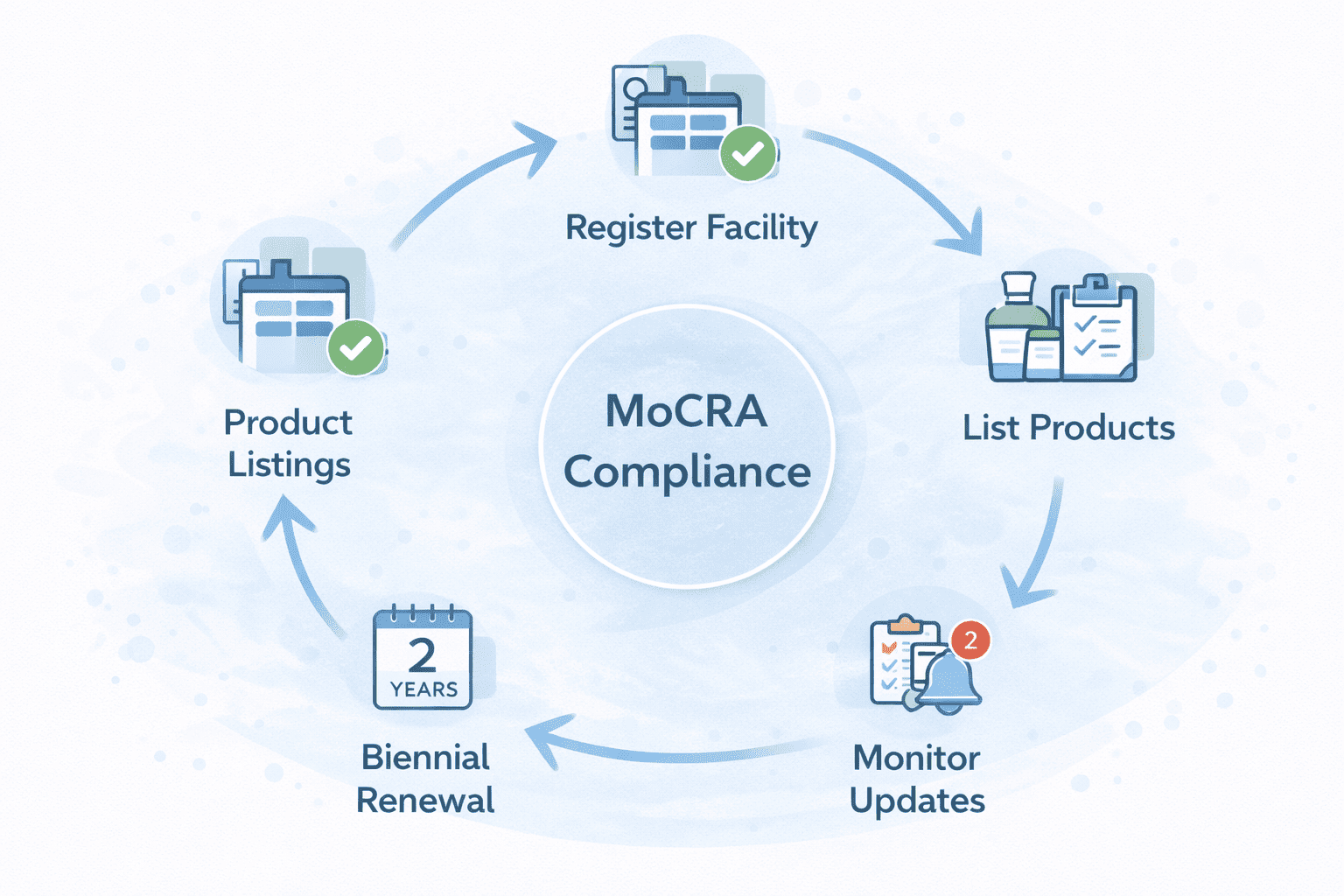
MoCRA is the Modernization of Cosmetics Regulation Act of 2022, which expanded FDA oversight of cosmetics. The law introduced requirements such as cosmetic facility registration that is renewed every two years, and cosmetic product listings that include ingredient information and are updated annually.
MoCRA Filings is a done-for-you paperwork workflow. It includes structured collection of facility and SKU information, QA for completeness and consistency, submission support through the appropriate FDA pathway, and delivery of a proof bundle with a tracker and update calendar.
At a glance
Renewed every two years (biennial).
SKU-level listing with ingredient information.
Product listings updates supported with common change triggers.
Share-ready submission evidence + tracker + update calendar.
MoCRA filings involve more than entering information into a portal. Facility details, product listings, ingredient information, and documentation must be organized correctly so submissions move forward without unnecessary delays.
BeyondBound uses a structured workflow that keeps the process clear from the very beginning. Information is collected in an organized format, reviewed for consistency, and prepared so filings can move forward with confidence.
The result is a documented process that leads to completed filings and a share-ready proof bundle, along with a clear tracker for future updates and renewal timelines.
A structured filing workflow built for real SKUs, real launches, and real deadlines.
Facility, responsible person, SKU, and ingredient information is collected in a clean and organized format. This prevents missing data and inconsistent naming that can cause confusion later in the process.
Instead of uploading scattered spreadsheets or guessing which fields matter, you move through a structured intake that prepares everything for filing from the start.
Before submission, the collected information is reviewed for completeness and internal consistency. This includes checking SKU names, ingredient entries, facility details, and alignment between documents.
The goal is to reduce preventable delays and avoid unnecessary back-and-forth once the filing process begins.
With information verified, support is provided through the appropriate FDA filing pathway. The process stays documented and organized so each step is clear and traceable.
You are not left navigating the portal alone. The workflow stays structured from intake through confirmation.
Once filings are completed, confirmations are packaged into a share-ready proof folder. This includes submission evidence, a filing tracker, and an update calendar.
The result is documentation you can store internally, provide to retailers or platforms, and reference for renewals and annual listing updates.

BeyondBound provides clear guidance and proactive support for MoCRA facility registration and cosmetic product listing through a documentation-first workflow that gives brands more clarity, better organization, and a cleaner path to submission. Filing evidence, tracking, and update visibility are built into the process so compliance does not become a last-minute scramble.
This approach does not simply point you to a portal. It provides a more dependable way to get filed, keep proof organized, and stay current as products and requirements evolve.
Taking you to "Filed" with structure, clarity, and documentation discipline.
You are not guessing which information matters or how it should be organized. The intake process is structured so facility details, SKU information, and ingredient data are collected in a way that aligns with filing requirements from the start.
That clarity reduces confusion before submission even begins. It also helps prevent missing details, inconsistent records, and last-minute rework that slows down progress later.
Incomplete or inconsistent information often causes delays and repeated follow-ups. By reviewing data before submission, many preventable issues are identified early and resolved before they interrupt the filing process.
This keeps filings moving forward instead of restarting the process midstream. It also reduces avoidable emails, correction cycles, and stalled timelines caused by missing or unclear information.
Filing is not just about submitting information. It is about having confirmations and records that stand up to internal review, retailer requests, and platform inquiries without scrambling for proof later.
The proof bundle organizes confirmations and tracking into something practical and shareable. It is more than a screenshot of a portal page. It is documentation you can reference with confidence.
MoCRA requirements do not end after submission. Facility registration renewals and product listing updates require ongoing attention, and missing timelines can create compliance gaps over time.
Renewal reminders and structured tracking help prevent missed deadlines. This ensures updates are handled proactively instead of becoming urgent issues right before renewal windows close.
Administrative filing support stays focused on what is required for submission. If something falls outside scope, such as potential claim or category conflicts, it is flagged early rather than filed blindly.
That discipline protects your process and prevents avoidable problems. It also keeps responsibilities clear so decisions are made intentionally, not under pressure after submission.
Cosmetic brands often file during busy launch cycles where teams are juggling multiple priorities. Clear communication and structured next steps keep the filing process manageable and predictable.
You always know what is needed next and what stage the filing is in. That visibility helps teams plan around deadlines without losing momentum during product launches.
Everything you need to reach "Filed" and stay current.
You receive a structured, share ready proof folder that contains your submission confirmations and organized filing records. This documentation is prepared in a way that supports internal compliance tracking and can be provided to retailers, platforms, or partners if requested. Instead of relying on portal screenshots, you have clear evidence of what was filed and when.
You receive a clean SKU tracker that shows which products were filed, the date of submission, and the status of each listing. As product lines grow or formulas evolve, this tracker becomes a central reference point. It reduces confusion and gives you a reliable record of what is current and what may require updates.
You receive a renewal and update calendar that outlines facility registration renewal timing and annual product listings obligations. The calendar also highlights common change triggers that may require updates. This helps prevent missing renewal deadlines and keeps compliance visible over time.
You receive organized administrative filing support from intake through submission confirmation. Information is reviewed for completeness and consistency before filing, and any obvious gaps are flagged early. The result is a cleaner path to completion and a documented workflow that supports long-term record keeping.
We manage facility registration workflows for brands and manufacturers that require accurate documentation and a clear submission path. This includes organizing required data, preparing filings, and maintaining renewal-ready records so biennial deadlines are not missed or delayed.
We support structured product listing workflows for brands with multiple SKUs, variants, and evolving product lines. SKU and ingredient details are standardized for consistency, making submissions easier to complete and simpler to maintain over time.
Filing delays often stem from inconsistent naming, scattered records, or incomplete ingredient data. We bring structure to SKU and ingredient information so submissions move forward smoothly without preventable confusion or rework.
Formula adjustments, packaging updates, or label revisions frequently require listing changes. We help track update triggers and maintain a filing structure that supports ongoing revisions and annual compliance needs.
Retailers and distribution platforms often request confirmation beyond initial filings. We organize submission proof and supporting documentation in a clear, reference-ready format that is easy to retrieve and share when needed.
Administrative support remains focused and structured, while obvious conflicts are identified early. This reduces avoidable filing issues caused by category misalignment, claim inconsistencies, or incomplete information.
MoCRA is not a one-time task. Updates are tracked so you don’t miss the next cycle.
Facility registration renewals occur every two years. We help keep the renewal cycle visible so deadlines do not get missed as teams shift focus back to launches and operations.
Product listings require annual review and updates. You receive reminders tied to your filing records so updates stay consistent and easy to manage over time.
As SKUs evolve, updates may be required. We help flag common change triggers so filings stay aligned with your current catalog and product information.
Your filings, confirmations, and update cycles stay organized in a central tracker. This gives you a clear reference point instead of scattered files and portal history.
Send your details and we'll guide your next steps. Someone from our team will contact you within 1 hour (during business hours), or at your requested time.
1980 Festival Plaza Dr. Suite 300, Las Vegas, NV 89135
info@beyondbound.org
(646) 217-0403