FDA registration and listing follow a consistent annual schedule. Missing the annual window can slow down shipping, onboarding, and partner approvals. BeyondBound supports the workflow by organizing required information, guiding portal steps, and packaging clean confirmation evidence for future reuse.
Renewal windows, user fees, and portal requirements create friction for many teams. A structured intake and quality check help reduce preventable back and forth and keep annual maintenance predictable.
Included with each annual cycle are U.S. Agent documentation when required, confirmation evidence, a registration and listing tracker, and a renewal timeline with a change control checklist.
We Support:
Stay organized year over year with documentation that is easy to reuse and easy to show when proof is requested.

Many foreign manufacturers and importers must designate a U.S. Agent as a U.S. based contact for FDA communications. U.S. Agent information is submitted as part of foreign establishment registration and should remain current year over year.
Medical devices and drugs operate on annual cycles that require ongoing attention. For devices, establishment registration must be renewed each year between October 1 and December 31, and listing information must be reviewed during the same period. For drugs, establishments renew registration annually between October 1 and December 31, and drug listing certification is completed during the annual certification period.
FDA U.S. Agent + Annual Registration & Listing is a done for you paperwork workflow. It includes structured collection of establishment and product information, QA checks for completeness and consistency, guided portal steps, and delivery of a proof bundle with a tracker and renewal timeline.
At a glance
U.S. Agent coverage is available when required, with acceptance documentation and a simple communications routing protocol.
Registration and listing review cycles are supported with a repeatable process aligned to the October through December window.
Device listing support and drug listing maintenance support are provided, including annual certification expectations during the annual period.
Confirmations are packaged with a tracker and a change control checklist so year over year updates stay clean.
Foreign establishments that sell medical devices or drugs in the United States often need a designated U.S. Agent and must maintain annual FDA registration and listing records. Keeping these details current requires consistent documentation and attention to yearly renewal timelines.
BeyondBound supports the administrative workflow by organizing establishment information, confirming listing details, and preparing records in a format ready for submission and future updates.
Each cycle concludes with a documented proof bundle, a tracker of registrations and listings, and a clear timeline so annual maintenance remains predictable year after year.
A structured annual workflow built for real establishments, real products, and real renewal deadlines.
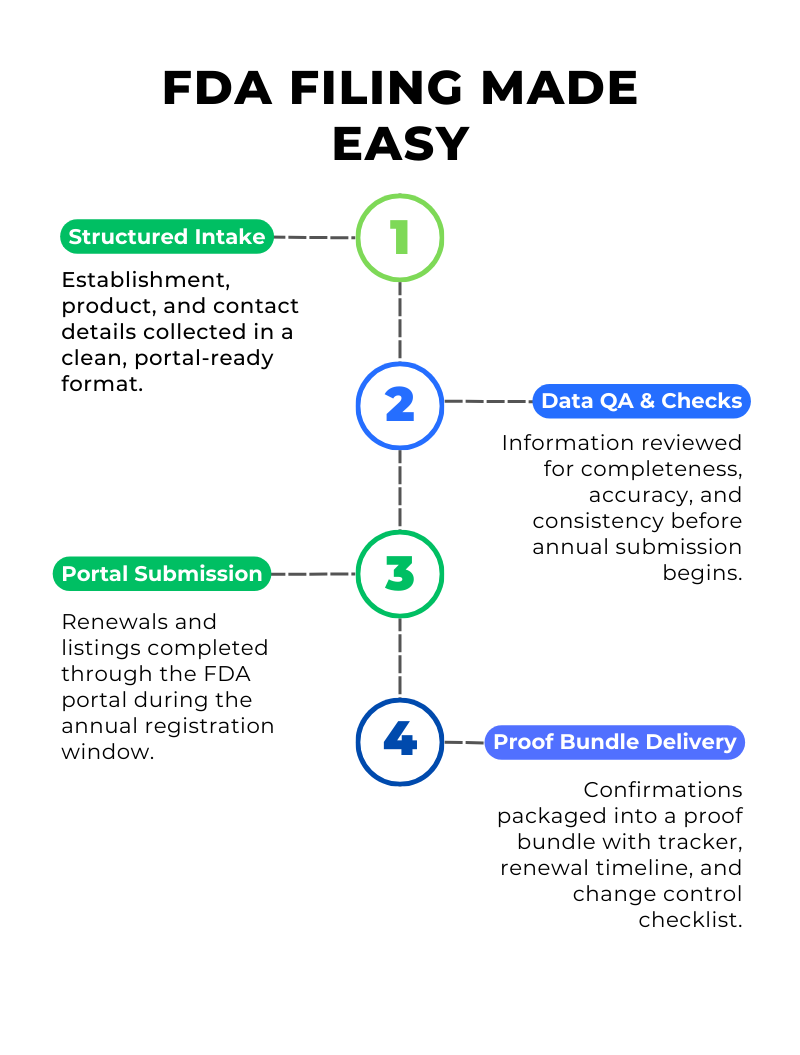
Establishment details, ownership contacts, product catalog information, and required identifiers are collected in a clean and organized format. This reduces missing fields and inconsistent naming that can create delays during registration and listing steps.
Collected information is reviewed for completeness and internal consistency. This includes establishment address and contact alignment, product listing details, and portal ready formatting. Obvious gaps are flagged early so the workflow does not stall during the annual window.
Support is provided through the appropriate FDA portal steps based on the registration and listing pathway. The workflow accounts for annual timing, user fee sequences when applicable, and documentation discipline so each step is traceable and easy to confirm.
Once annual submissions are completed, confirmations are packaged into a share ready proof folder. You receive evidence, a tracker view, and a renewal timeline with change triggers so year over year updates remain clean and predictable.
BeyondBound supports deadline-driven FDA workflows with a documentation-first approach that keeps annual registration and listing work clear, organized, and easier to maintain over time. Instead of treating each cycle like a new project, the service creates a repeatable system with confirmation evidence, tracking, and renewal visibility built in.
This approach does not simply point you to a portal. It gives foreign establishments a more reliable way to manage U.S. Agent support, annual filings, and ongoing updates without last-minute scrambling, scattered records, or unnecessary rework.
Taking you to "Completed" with structure, clarity, and documentation discipline.
Annual FDA registration and listing work follows strict timelines. The intake process is structured so establishment details, listing information, and U.S. Agent requirements are gathered in a portal ready format from the start.
That clarity reduces confusion and prevents rebuilding records each year. Information stays organized in a way that supports smooth submission and future renewals.
Incomplete or inconsistent information often creates delays during the October through December renewal window. By reviewing data before submission, many preventable issues are identified and corrected early.
This keeps annual maintenance moving forward without restarting the workflow. Teams avoid unnecessary corrections and last minute pressure during busy cycles.
Annual submissions are only valuable when confirmations are organized and easy to reference. The proof bundle packages evidence in a share ready format that supports compliance tracking and partner requests.
You maintain clean records of what was submitted and when. Documentation remains accessible for audits, renewals, and distributor onboarding.
Renewal obligations continue after the initial submission is complete. Registration renewals, listing reviews, and certification expectations require attention each year to stay aligned with FDA requirements.
Tracking and reminders help prevent deadlines from quietly passing. The process stays proactive rather than reactive as renewal periods approach.
Administrative support stays focused on filings and communications routing. Legal advice, regulatory interpretation, and technical opinions are not provided as part of the service.
If something falls outside scope, it is identified early and communicated clearly. This keeps responsibilities defined and prevents misunderstandings later.
Teams often need submission proof for onboarding, distributor requests, customs inquiries, or platform requirements. Clear communication and structured next steps help keep the workflow manageable.
You always know what is required next and what stage the filing is in. That visibility helps teams meet shipping and partner deadlines with confidence.
Everything you need to complete annual filings and stay current year over year.
You receive a structured, share ready proof folder containing confirmations, portal evidence, and organized filing records. This documentation supports internal compliance tracking and can be provided to partners, distributors, or platforms if requested.
You receive a clean tracker showing establishment details, registrations, listing items, submission dates, and annual status. As products and contacts evolve, this becomes the central reference point for what is current and what needs updating.
You receive a renewal timeline aligned to the annual October through December window, plus a change control checklist for common update triggers. This helps prevent annual filings from becoming a scramble and keeps updates consistent.
You receive organized administrative filing support from intake through confirmation. Information is reviewed for completeness and consistency before submission, and obvious gaps are flagged early to keep the workflow moving.
We provide U.S. Agent coverage when required, including formal acceptance documentation and clear communication routing so FDA correspondence is properly received and organized.
This ensures regulatory notices are not missed and responses remain structured. Communication stays centralized and easy to manage throughout the registration cycle.
Annual timelines are supported with a repeatable renewal process aligned to the October through December window. The workflow is organized in advance for predictable and manageable submissions.
Clear milestones and preparation steps reduce last minute pressure. The process remains steady and controlled rather than rushed at year end.
We assist with device establishment registration and annual listing review to ensure required submissions are complete and aligned with FDA expectations.
This keeps the registration cycle coordinated from submission through confirmation. Annual requirements are handled in a consistent and organized manner.
Drug establishment renewal support is provided along with annual listing certification assistance during the required certification period. Documentation is prepared carefully for accurate submission.
Records are packaged clearly for reuse and future reference. This helps maintain continuity across certification cycles and regulatory reviews.
Common update triggers are tracked, including new products, discontinued items, and address or contact changes. This helps ensure listings remain current and aligned with regulatory records.
Change control keeps year over year updates clean and organized. Adjustments are documented in a way that supports both internal tracking and future submissions.
Completion evidence is compiled into a share ready proof folder designed for onboarding, distributor requests, and annual renewals. Documentation is organized for clarity and accessibility.
Proof remains easy to retrieve when needed for audits or partner reviews. Records are maintained in a format that supports ongoing regulatory confidence.

Everything packaged clearly so it is obvious what was completed and what to update next year.
Evidence packaged cleanly and labeled clearly.
What was registered and listed, and when.
Annual window reminders built into the deliverables.
New products, discontinued items, contact changes.
The goal is to make next year easier by keeping proof, trackers, and change triggers organized now.
FDA registration and listing are not one time tasks. Annual cycles are tracked so you do not miss the next window.

Annual renewals occur on a fixed timeline. We keep the cycle visible so deadlines are not missed as teams shift focus back to operations and shipping.
Many workflows depend on the correct order of steps, portal access, and user fee timing. The process is structured so steps stay aligned and traceable.
Updates may be required when products change, contacts change, or establishments change. We help flag common triggers so annual maintenance stays consistent.
Your confirmations, tracker, and renewal timeline stay organized in a central proof bundle so you can answer partner requests quickly without digging through portal history.
Send your details and we will guide your next steps. Someone from our team will contact you within 1 hour (during business hours), or at your requested time.
1980 Festival Plaza Dr. Suite 300, Las Vegas, NV 89135
info@beyondbound.org
(646) 217-0403